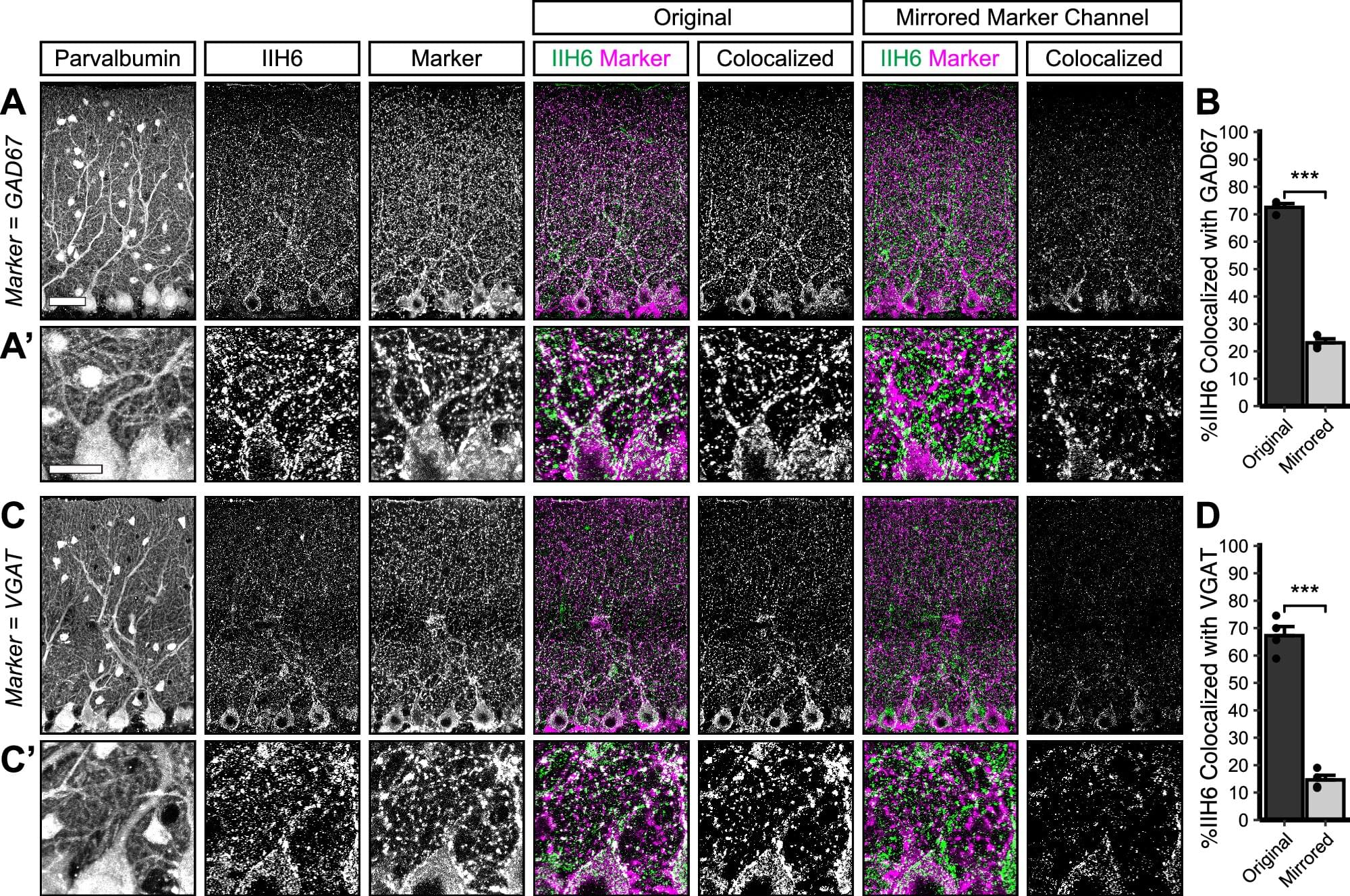
Scientists have uncovered how a protein helps build and maintain vital brain connections, providing insights into the neurological problems experienced by people with a rare form of muscular dystrophy known as dystroglycanopathy.
The research conducted at Oregon Health & Science University and published in Communications Biology reveals that the protein Dystroglycan plays a critical role in forming and maintaining connections between nerve cells in the cerebellum—the part of the brain responsible for movement coordination and motor learning.
In people with dystroglycanopathy, genetic mutations in the protein affect not only muscles but also the brain. The condition is a type of congenital muscular dystrophy, a group of inherited disorders that appear at birth or in early infancy.