While E. Josie Clowney would never suggest that neuroscience is simple, a new study by her team at the University of Michigan could drastically reduce complexity in future studies. Their work focused on instinctual behaviors in fruit flies, but it has the potential to accelerate work to better understand the neurobiology that underlies behavior and decision-making in mammals, including humans.
The research establishes a new way to understand neurons, their connectivity and the behaviors they control. Within this new framework, the researchers can circumvent the conventional approach of considering each type of neuron individually and instead focus on groupings defined by shared structure and by two sets of regulatory genes. The work is published in the journal Nature.
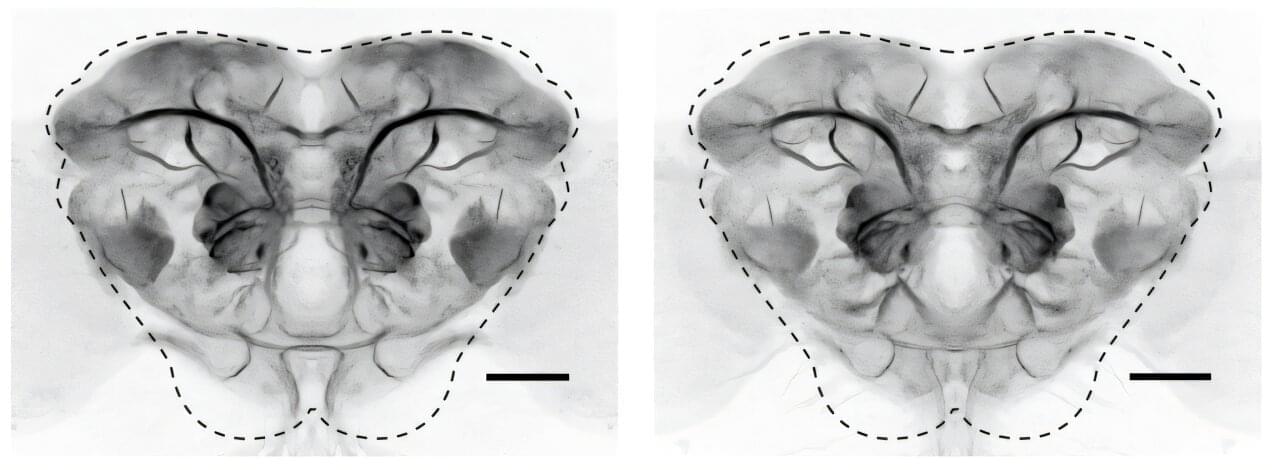
While there are more than 8,000 kinds of neurons in the fruit fly cerebrum —the part of its brain where instinctual behaviors are hardwired—there are less than 200 major structural groups, or ground plans. Led by Najia Elkahlah, who recently defended her doctoral thesis in the Clowney lab, the team’s discoveries revealed how these ground plans get set up. There is a sort of order or hierarchy, where one set of genes coordinates the formation of the ground plan, and the other set produces small differences in shape and connectivity among neurons within each ground plan.