Tang, L., Singh, S.C., Wei, R. et al. Light Sci Appl 15, 246 (2026). https://doi.org/10.1038/s41377-026-02315-4

Tang, L., Singh, S.C., Wei, R. et al. Light Sci Appl 15, 246 (2026). https://doi.org/10.1038/s41377-026-02315-4
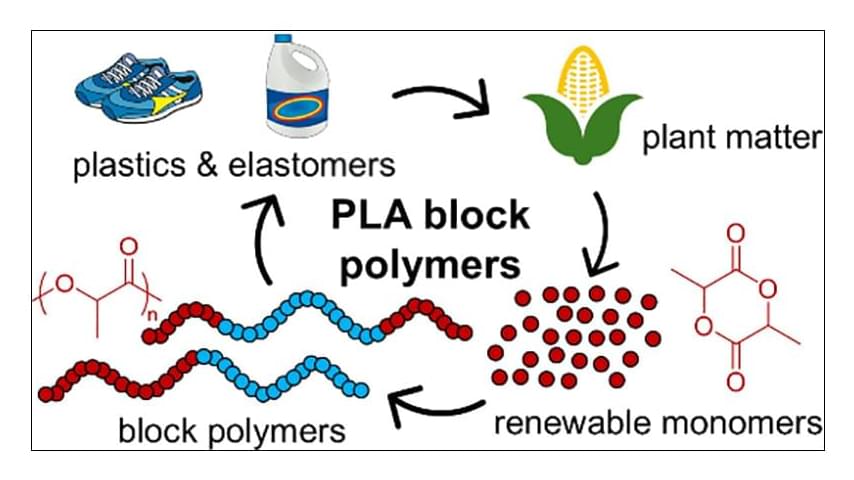
Block polymers present an almost endless realm of possibilities to develop functional materials for myriad applications. The established self-assembly of block polymers allows researchers to access properties that are inaccessible in homopolymers. However, there is a need to develop more sustainable options than the current commodity block polymers. Derived from renewable resources and industrially compostable, poly(lactide) (PLA) is at the forefront of technological advancements in sustainable block polymers. Its material properties including high stiffness, relatively high glass transition temperature, and semicrystallinity in isotactic versions lend themselves to many applications, and its ease of synthesis provides a well-established platform for developing high-performance materials. This Perspective highlights recent advancements associated with PLA-containing block polymers, including their syntheses, mesostructural considerations, and mechanical properties, from resilient elastomers to tough plastics. We also give our perspective on the subfield of PLA block polymers, our outlook on the future, and our assessment of exciting developments yet to come.

Freshwater from melting Antarctic glaciers may be influencing the Southern Ocean in ways scientists have largely overlooked. New research, published in Frontiers in Marine Science, has found that glacial meltwater is not confined to the ocean’s surface, as previously assumed, but can also be detected much deeper in coastal waters along the Western Antarctic Peninsula.
The findings suggest that meltwater from glaciers is being transported and stored tens of meters below the surface, where it could alter ocean circulation, affect the movement of heat and nutrients, and influence how the region responds to climate change.
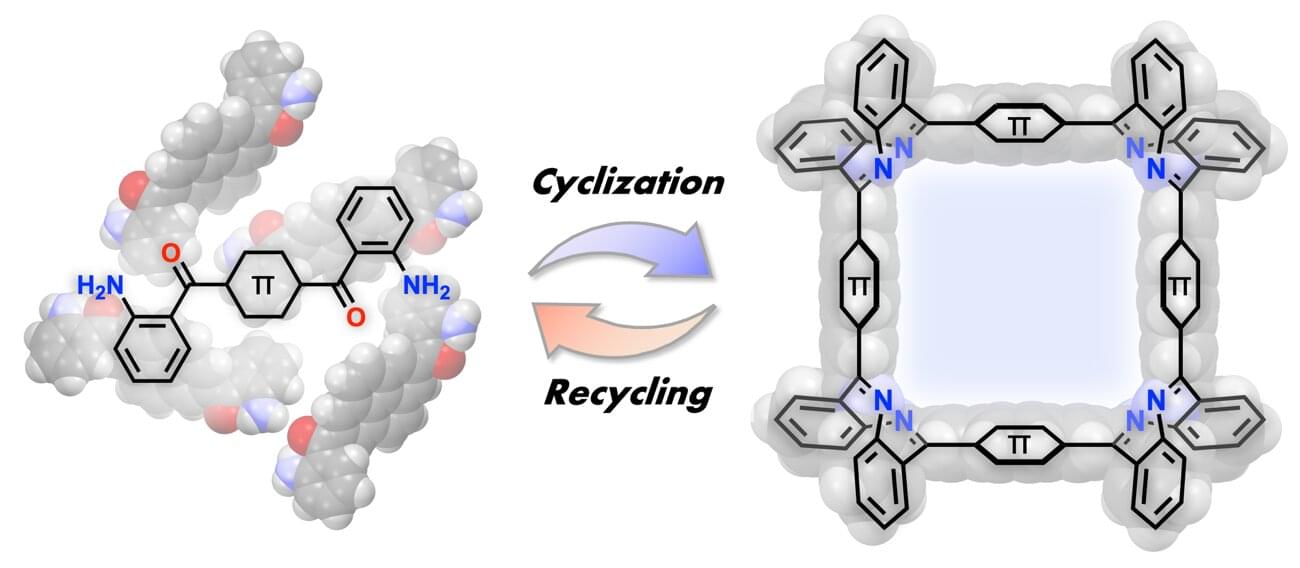
A research group has developed a new method for selectively synthesizing three-dimensional macrocycles,⁽¹⁾ in which four panels are arranged in a square, by connecting planar π-conjugated molecules⁽²⁾ at right angles.
This method is applicable to a wide variety of π-conjugated molecules and allows the size of the internal cavity to be designed. Furthermore, the resulting square macrocycles exhibit acid responsiveness, reversibly changing color under the action of a mild acid, while acid-mediated hydrolysis enables the starting monomers to be recovered in high yield—realizing a sustainable molecular synthesis that reverts to and regenerates the starting materials. The originality of this work lies in having a single imine bond play three roles: creating the shape, responding to stimuli and reverting back.
These research results were published in the Journal of the American Chemical Society on Monday, June 1, 2026. The team includes Associate Professor Yasutomo Segawa and Assistant Professor Takashi Harimoto at the Institute for Molecular Science (National Institutes of Natural Sciences) and the Graduate University for Advanced Studies (SOKENDAI).

Waves of light and sound interact to drive electronic and structural changes in a perovskite crystal. At the atomic scale, nothing is ever truly still. Materials that appear perfectly rigid and motionless to the naked eye are in fact swarms of vibrating atoms. This motion is generally random and uncoordinated, but with the right input, the atoms in certain materials will start to move together, vibrating in sync.
These collective vibrations are a form of sound called phonons, and when tuned just right, they can influence a material’s structure and behavior in dramatic and useful ways. Researchers are working to understand and control this effect to optimize material properties and even access hidden phases of matter.
Scientists at the U.S. Department of Energy’s (DOE) Argonne National Laboratory are using light to drive phonon activity in a class of materials called metal halide perovskites, whose customizable structures and photosensitivity hold promise for use in next-generation solar cells, advanced sensors and quantum information technologies.

Storm Dave, which swept across northern Europe over the Easter weekend, is an example of what new research from the University of Gothenburg has revealed. Spring storms forming over the North Atlantic have become more common than they were 80 years ago, and this is due to climate change.
In the Northern Hemisphere, storm seasons follow a seasonal cycle. Storms are weakest and least frequent in summer and most intense in winter. As a result of global warming, storm patterns and their course have changed, and several studies have indicated that winter storms appear to be occurring more frequently and with even greater intensity.

A recent expedition to Central Africa has uncovered dozens of new species.
In February, a team of 16 specialists from Africa and around the world visited the Lisima plateau in eastern Angola and conducted a biodiversity survey, through which they discovered dozens of species unknown to science, according to The Wilderness Project, which led the survey.
The organization dedicated to studying and protecting Africa’s freshwater wilderness announced the findings from the remote scientific expedition in the area — seen as one of Africa’s last great biodiversity blank spots — in a news release obtained by PEOPLE on Wednesday, June 3.

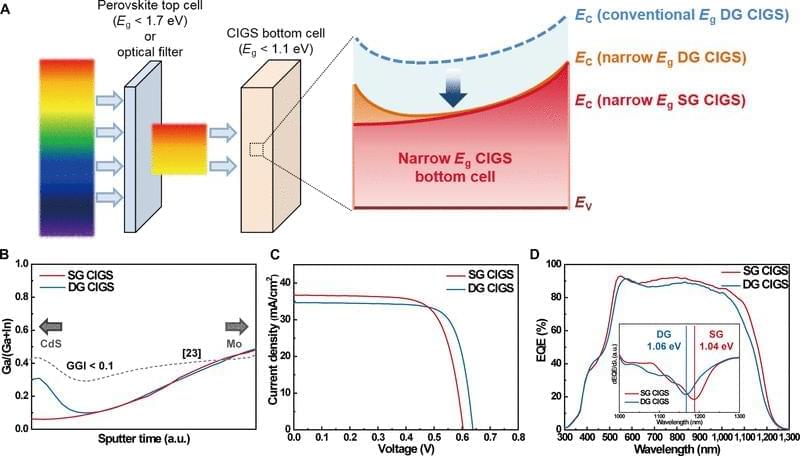
This study examined the potential of narrow-bandgap (Perovskite-based tandem solar cells are a promising photovoltaic (PV) technology to exceed the Shockley–Queisser limit of single-junction solar cells. Perovskite/Si tandem solar cells have been intensively studied, demonstrating a record power conversion efficiency (PCE) of 34.6% [1]. In contrast, the certified record PCE of perovskite/Cu(In, Ga)Se2 (CIGS) tandem solar cells remains 24.6% with a reported efficiency of 24.9% [1, 2]. Theoretical calculations for double-junction tandem solar cells using a detailed balance model indicate that the bandgap (Eg) combinations of 1.12 eV (for a bottom cell) and 1.70 eV (for a top cell) or 0.90 to 1.04 eV (for a bottom cell) and 1.58 to 1.67 eV (for a top cell) can yield a maximum theoretical tandem efficiency [3, 4]. Wide-bandgap perovskite (with Eg equal to or greater than 1.7 eV) has been actively studied for tandem application with Si (Eg = 1.12 eV), the most successful solar cell technology to date as a bottom cell. However, previous studies have shown that wide-bandgap perovskite suffers from substantial open-circuit voltage (VOC) loss due to halide segregation [5], and the maximum PCEs of single-junction perovskite cells have been produced by perovskite with Eg between 1.52 and 1.63 eV [6– 8]. The bandgap of CIGS can be tuned between 1.01 and 1.68 eV by adjusting the Ga/(Ga+In) (GGI) ratio and through tuning of bandgap grading profile [9]. Employing a narrow-bandgap CIGS close to 1.00 eV as a bottom cell is advantageous to use the most efficient, conventional bandgap perovskite as the top cell. Therefore, unlike Si, the bandgap tunability of CIGS offers an opportunity for perovskite/CIGS to attain a greater ultimate performance than perovskite/Si tandem solar cells. Han et al. [10] introduced a thick indium-doped tin oxide (ITO) recombination layer to bury the intrinsic surface roughness of CIGS, followed by chemical mechanical polishing to prepare a smooth surface for the subsequent solution process of perovskite, attaining a certified PCE of 22.4%. Albrecht and coworkers have improved the PCE of perovskite/CIGS tandem solar cells by modifying the hole transport layer (HTL). In their earlier work, a NiOx/PTAA bilayer was utilized to form a uniform HTL on CIGS bottom cells. Recently, a self-assembled monolayer such as 2PACz and Me-4PACz was used, which can enhance the device performance of single-junction perovskite solar cell and its perovskite/CIGS tandem counterpart, achieving a certified PCE of 24.2% [2, 11 – 13].
Most recent studies on perovskite/CIGS tandem solar cells have focused on optimizing the perovskite top cell. In contrast, all CIGS bottom cells include an absorber with a double-graded (DG) bandgap profile optimized around the bandgap of ~1.1 eV. The DG bandgap profile has been adapted primarily for CIGS absorbers prepared by thermal evaporation, which has resulted in high-performing CIGS solar cells with PCEs up to 23.4% [14], and it has proven to be an effective strategy for enhancing performance, optimized for “single-junction” CIGS; however, it has not been determined whether DG would be the ideal configuration for tandem applications. Kim et al. [15] used single-graded (SG) CIGS with a bandgap close to 1.0 eV, where the band grading is only formed on the backside of the absorber. They employed dual alkali post-deposition treatment (PDT) with KF and CsF, demonstrating a CIGS solar cell with a PCE of 20.

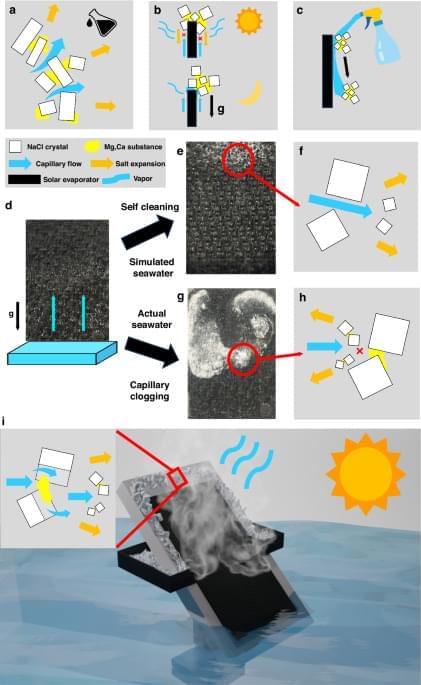
Scientists have developed a solar desalination system that turns seawater into drinking water without creating environmentally damaging brine. Special laser-textured metal panels use sunlight to evaporate water while automatically moving salt deposits away from the working surface, preventing clogging. The process was successfully tested with water from three oceans and can recover nearly all salts as solids. Those leftover materials could even become a source of valuable lithium for batteries.