Researchers surveyed over 300 physicians to get their take on leaving patients in suspended animation for future revival.


Join us for a deep-dive conversation with Dr Emil Kendziorra, CEO of Tomorrow.bio, as we explore the cutting edge of cryopreservation and medical biostasis.
Dr Kendziorra, who began his career in cancer research with a summa cum laude degree from the University of Göttingen, shares his remarkable journey from academic science to entrepreneurship, and ultimately to founding Tomorrow.bio — a company he considers his life’s work.
In this interview, he discusses the motivations behind his pivot from traditional longevity research to the frontier of cryomedicine, the operational and emotional lessons learned from cryopreserving over 20 human patients and 10 pets, and his vision for making this technology more accessible in the future.
Tomorrow.bio stands as Europe’s fastest-growing cryopreservation company, with over 800 members across 200+ cities and 45+ countries. The company offers both whole-body cryopreservation and brain-only preservation, using a transparent pricing model designed for long-term value.
With a recent €5 million Seed round and a mission to advance medical biostasis, Emil discusses the unique challenges and opportunities of operating in this space, the ethical considerations of a technology that may not achieve revival for decades, and his long-term vision for the field.
Key Points.

What counts as death? And who gets to decide?
In the summer of 2013, I traveled to Scottsdale, Arizona to visit the Alcor Life Extension Foundation, the world’s leading cryonics organization, founded in 1972. CEO Dr. Max More gave me a full tour of the facilities and walked me through the entire process: from the moment clinical death is declared, through controlled cooling and vitrification, to the cryo-tanks holding (at the time) 117 patients in long-term storage.
I also asked him, somewhat selfishly, whether my big bald head would fit comfortably in a neuro-patient container.
After the tour, Max sat down with me for a 25-minute conversation that covered:
Affordability and the real cost of membership Why minimizing cooling delays after clinical death is critical, and what long-distance members do about it Preserving pets, because of course people ask Chemical brain preservation as an alternative path The importance of protecting the neuron’s microtubules The case for an X Prize style competition to reduce tissue damage Where cryonics sits inside the broader transhumanist project.
My favorite line from Max, the one I still come back to:
Dr. Fahy is the Vice President and Chief Scientific Officer at 21st Century Medicine, Inc, and has co-founded Intervene Immune, a company developing clinical methods to reverse immune system aging. He was the 2022–2023 president of the Society for Cryobiology. Dr. Fahy is the lead author of a recent paper, “Ultrastructural and Histological Cryopreservation of Mammalian Brains by Vitrification” – the main topic of our conversation.
In December of 2014, I worked with Dr. Fahy to cryopreserve Dr. Stephen Coles under special conditions, with his permission to extract brain samples and test them for preservation quality. We did not know what the results would be. If bad, that would be discouraging for cryonics. In fact, the results were excellent, as Dr. Fahy details.
We discuss the Coles case and the results of the cerebral cortical biopsy. The paper includes results from rabbit brains. We also discuss the relative resilience of the brain compared to other organs when it comes to fracturing; how cryoprotectants prevent ice formation even when the blood-brain barrier remains closed; whether biostasis organizations should be using blood-brain barrier opening agents; Dr. Fahy’s thoughts about chemical preservation and the role of a combination of cryo an chemo, known as aldehyde-stabilized cryopreservation (ASC), and more.
Our speaker this month is Jordan Sparks with the Sparks Brain Preservation organization in Oregon. Our event is in ZOOM Only, no in person meeting this month, meeting ins ZOOM on Thursday, April 30th, opening at 6:00 PM for our social hour, with the main event starting at 7:00 PM Eastern Time Jordan will tell us about his project, which was formerly the Oregon Brain Preservation, and before that Jordan formed Oregon Cryonics. This is an entirely different type of bio-stasis then cryonics. Their stated goal is to preserve the structure of the entire brain at a fine ultrastructural level. This includes the synaptic architecture as well as detailed molecular information such as protein post-translational modifications, cellular epigenetic patterns, and subcellular distributions of molecules.
Cryonics and biostasis have been little known and even less practiced in South America. Diego Zarco is working to change that in Mexico. Diego started Criogenia Avanzada in Mexico City and has been developing and funding the organization, preparing to offer SST (standby, stabilization, and transport) services with storage to be done outside the country.
In this discussion, Max More asks Diego how he become interested in cryonics, what his organization does, where Criogenia Avanzada is located in the city and how it will reach patients quickly, and how his experience in the music industry prepared him for the difficult work of making biostasis work in practice.
Imagine a world where anyone who needs a bone marrow transplant can get one — on demand. No more desperate donor searches or deadly delays. Kevin Caldwell, Co-Founder & CEO, Ossium Health.
Bone marrow transplants have always depended on finding the right donor at the right time. But what if bone marrow could be stored, shipped, and used on demand—just like a drug? That’s exactly what Ossium Health is now showing in human clinical data.
Kevin Caldwell is the Co-Founder, CEO, and President of Ossium Health (https://ossiumhealth.com/), a clinical-stage bioengineering company pioneering off-the-shelf, cryopreserved bone marrow therapies derived from deceased organ donors.
Under Kevin’s leadership, Ossium has developed a novel platform designed to solve one of the most persistent challenges in transplantation medicine: timely access to compatible bone marrow for patients with life-threatening hematologic malignancies such as Acute Myeloid Leukemia. The company’s approach enables on-demand delivery of viable marrow cells, bypassing the logistical and biological constraints of traditional donor matching and scheduling.
Since its founding, Kevin has scaled Ossium from an early-stage startup into a clinical-stage company with a robust network of over 50 strategic partnerships across supply, clinical development, and commercial channels. He has led multiple financings and secured a landmark contract with the Biomedical Advanced Research and Development Authority, validating Ossium’s relevance to national health preparedness and biomanufacturing resilience.
Year 2025
Cryopreserving the adult brain is challenging due to damage from ice formation, and traditional freezing methods fail to maintain neural architecture and function. Vitrification offers a promising alternative but has not been surveyed in the brain. Here, we demonstrate near-physiological recovery of the adult murine hippocampus after vitrification of brain slices and of the whole brain in situ. Key features of the hippocampus are preserved, including structural integrity, metabolic responsiveness, neuronal excitability, and synaptic transmission and plasticity. Notably, hippocampal long-term potentiation was well preserved, indicating that the cellular machinery of learning and memory remains operational. These findings extend known biophysical limits for cerebral hypothermic shutdown by demonstrating recovery after complete cessation of molecular mobility in the vitreous state. This suggests that the brain can be arrested in time and then reactivated, opening avenues for potential clinical applications.
Significance Statement While the brain is considered exceptionally sensitive, we show that the hippocampus can resume normal electrophysiological activity after being rendered completely immobile in a cryogenic glass. The work extends known biophysical tolerance limits for the brain from the hypothermic to the cryogenic range and establishes a protocol for its long-term storage in a viable state.
The authors have declared no competing interest.
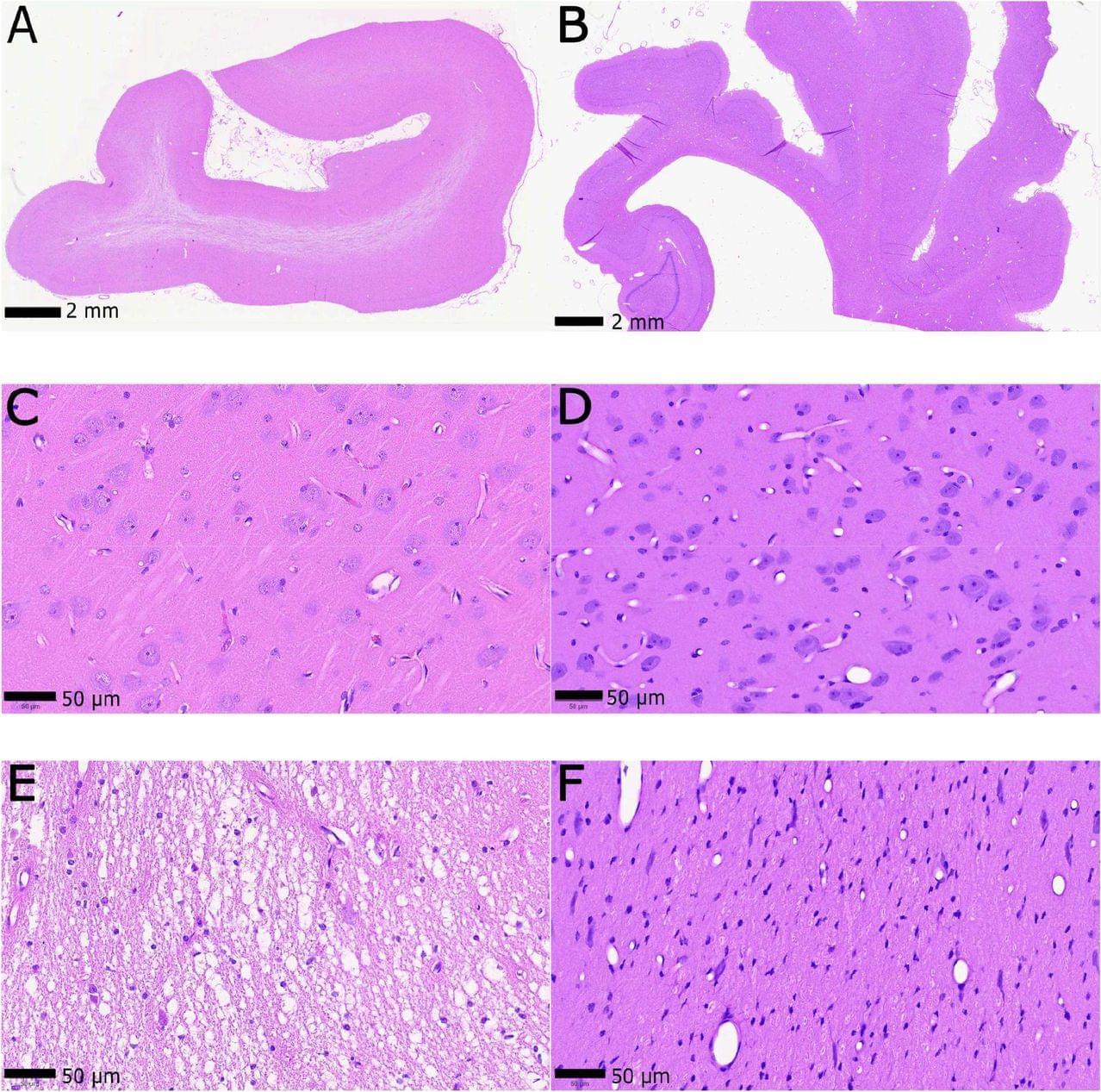
Ultrastructural Preservation of a Whole Large Mammal Brain (bioRxiv, 2026) ⚠️ Preprint – not yet peer-reviewed.
A 2026 preprint builds on over a decade of brain preservation research, demonstrating that whole mammalian brains (pigs) can be preserved with remarkable structural fidelity under near–real-world, end-of-life conditions.
The study refines aldehyde-stabilized cryopreservation (ASC)—a technique previously recognized by the Brain Preservation Foundation. This method combines chemical fixation (aldehydes), cryoprotectants, and controlled cooling to prevent ice damage and preserve neural structure at the nanoscale. — What the study shows.
Whole pig brains preserved with intact cellular and synaptic architecture.
Preservation remains viable even with delayed postmortem intervals (~10 minutes)
Tissue remains perfusable and structurally stable after fixation.
Protocol moves toward clinically realistic implementation, not just lab conditions.