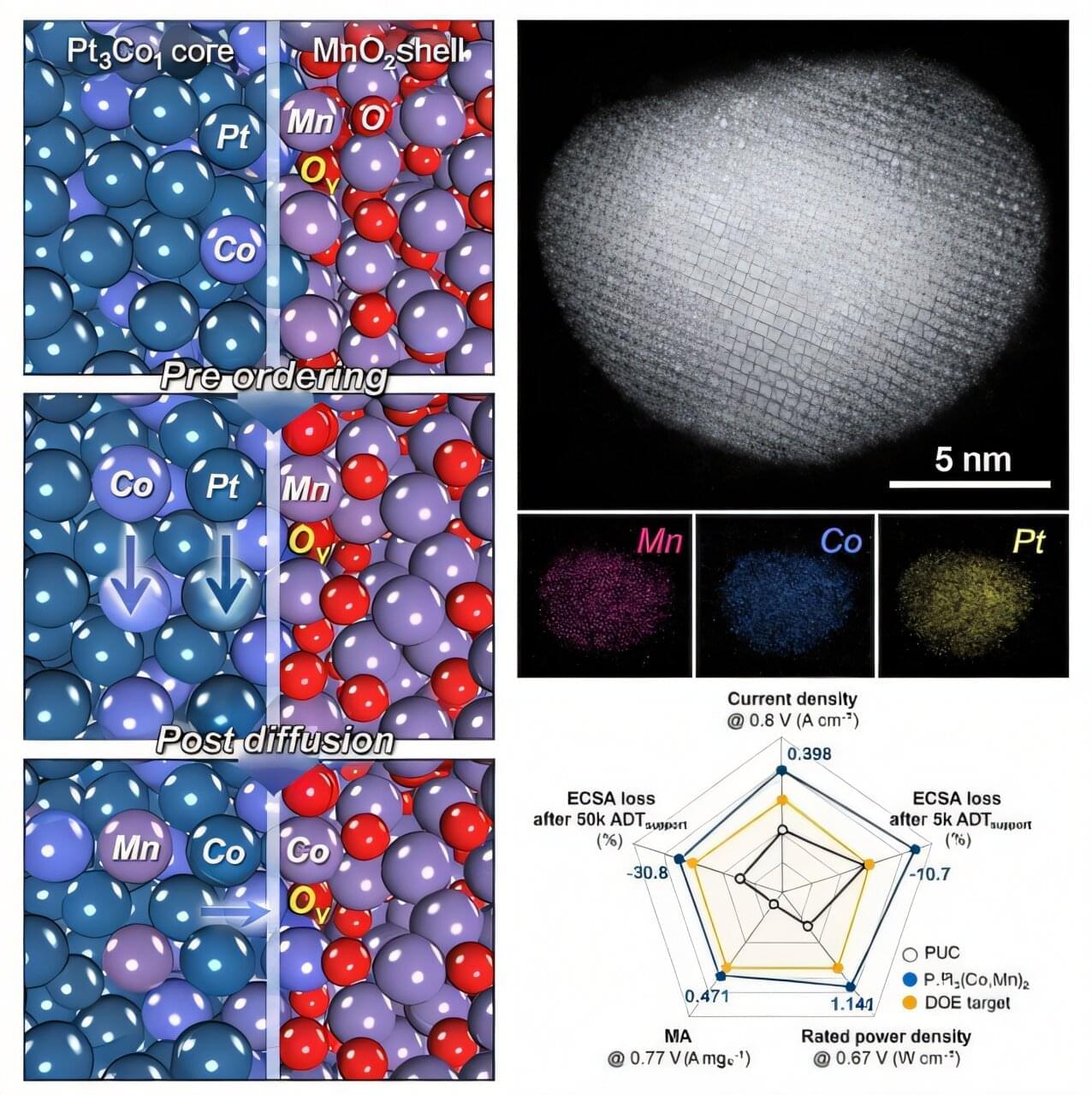
A robotic metamaterial shows that the odd mechanics of active solids depend on how the active constituents connect across the system.
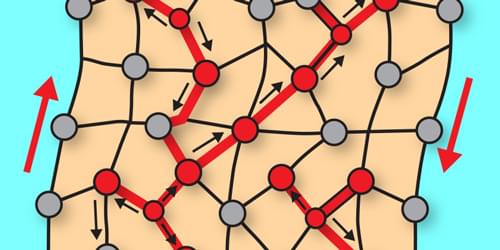
Active materials, composed of microscopic constituents that continuously inject motional energy into the system, can exhibit odd mechanical responses, such as stretching vertically when sheared horizontally. Such properties can be used to make materials that can spontaneously crawl or roll over a difficult terrain [1]. One might naively think that these desirable odd responses could be increased by making the components more active. Jack Binysh of the University of Amsterdam and his colleagues now find that this doesn’t always work [2]. The researchers show that in active solids a collective response only emerges when system-spanning connective networks are formed among the individual constituents of the system. Without such networks, the effects of microscopic activity remain confined locally and the macroscopic response disappears.
An active solid is, fundamentally, an elastic lattice made up of self-driving constituents. Examples include robotic lattices composed of motorized units [1, 2], magnetic colloidal crystals [3], and chiral living embryos [4]. The active solids that Binysh and his colleagues examined are examples of nonreciprocal active solids, meaning that the interactions between elements are directional. Interactions may become directional when individual constituents process information about their neighbors. Such nonreciprocal interactions arise in a wide range of settings. In robotic metamaterials, local control loops impose directional responses on adjacent mechanical units [1]. And in living chiral collectives, hydrodynamic flows allow rotating embryos to exchange momentum with the surrounding media [4].