During intensive cancer treatment, children, adolescents and young adults are exposed to numerous toxicities and psychosocial stressors that can cause psychosocial distress and impair mental health. The maintenance or rapid recovery of mental health during and after exposure to significant stressors has been defined as resilience. To date, resilience research has focused primarily on cross-sectional assessment of specific, trait-like resilience factors and concepts in long-term survivors of childhood cancer, typically omitting the influence of context-specific biopsychosocial stressors and resilience dynamics throughout treatment. Little is known about outcome-based resilience and mental health resources in childhood cancer patients undergoing cancer treatment. In addition, specific instruments for age-appropriate assessment of resilience in childhood cancer patients are lacking. To address this gap, within the EU Horizon 2020-funded FORTEe project, we developed a novel self-report instrument for longitudinal assessment of resilience in children, adolescents, and young adults with cancer, featuring age-appropriate items tailored to their specific contexts.
An interdisciplinary team of psychologists, psychiatrists and pediatric oncologists developed an age-appropriate self-report instrument to assess resilience longitudinally in children, adolescents, and young adults undergoing cancer treatment. Following current resilience research frameworks, resilience is defined as the ratio of changes in mental health problems to stressor exposure. Accordingly, the measure comprises two domains: mental health problems (anxiety, depression, distress, fatigue) and stressor exposure (daily hassles, cancer-related stressors), with stressors rated for both frequency and intensity.
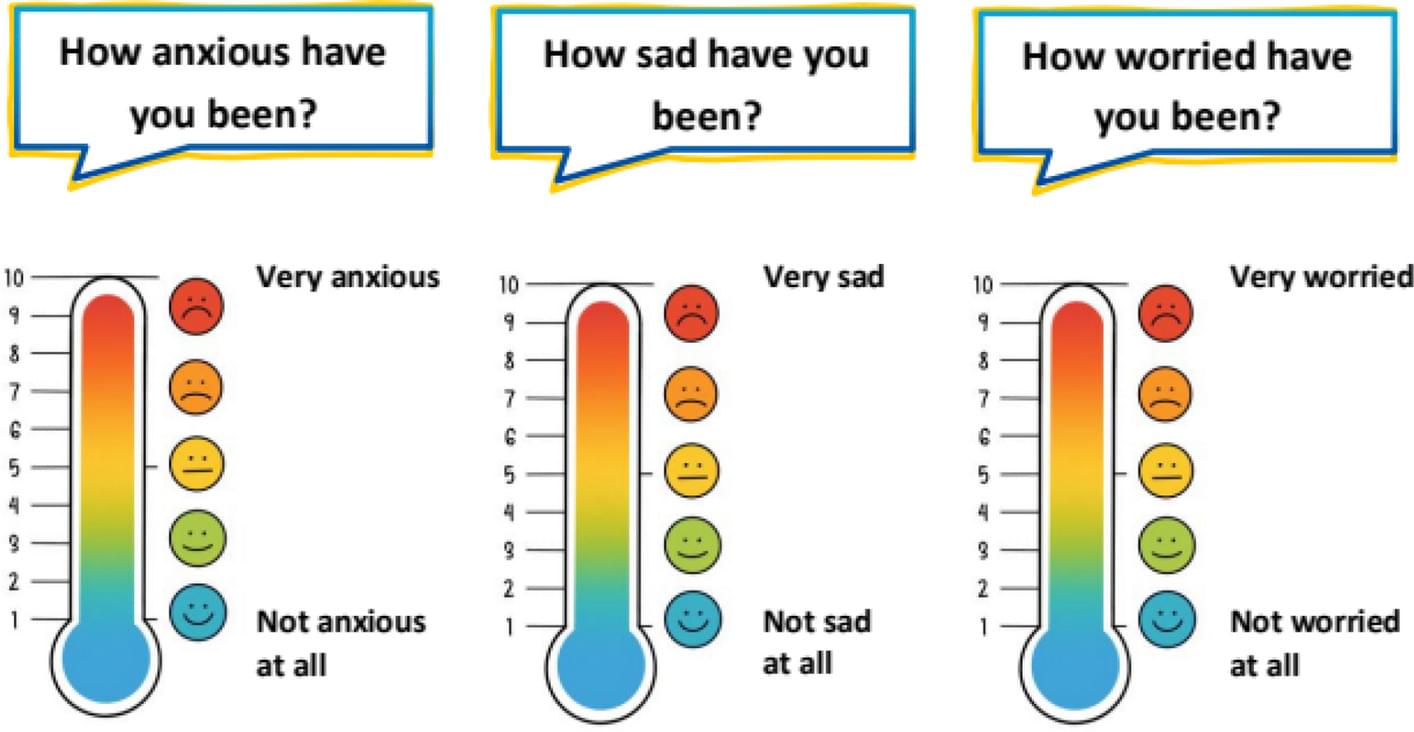
The Mainz Resilience Assessment in Childhood Cancer (MRAcc) consists of three age-specific versions (children 5–11 years, adolescents 12–17 years, young adults 18–21 years), each including the sections: ‘Emotions & Distress’, ‘Fatigue’, and ‘Situations & Experienced Stress’. It is available in German and English and uses either five-point-Likert scales or visual analogue scales presented as thermometers.