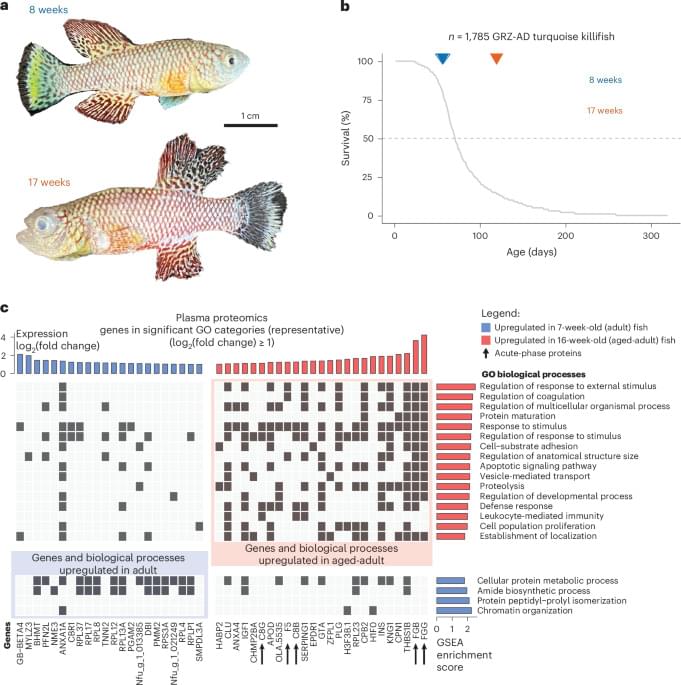
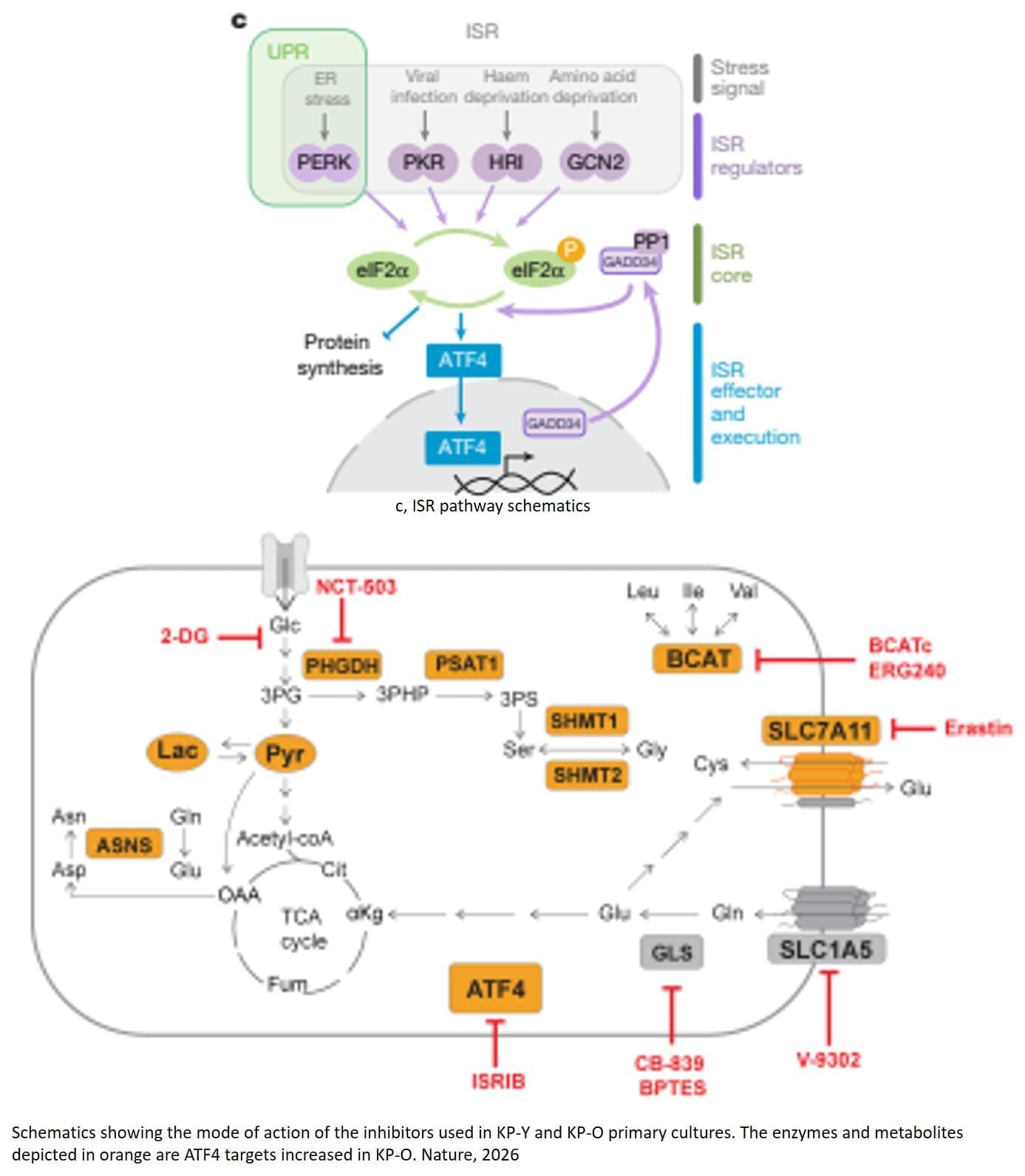
The study shows how aging alters the biology of lung cancer and makes tumors more prone to spreading. The researchers identified a molecular signaling pathway, a complex chain of reactions and interactions, in which a specific stress-response protein, ATF4, plays a central role.
Under normal physiological conditions, ATF4 acts as a hub for the “integrated stress response” that responds to events such as nutrient deprivation, viral particles, or the accumulation of misfolded proteins, activating protective and corrective responses, explains the author.
“In older patients, this stress response is hijacked by the tumor, allowing cancer cells to reprogram their metabolism. The tumor does not grow faster, but this metabolic rewiring enables the cancer cells to spread and form metastases in other parts of the body,” the author says.
Tumors from older individuals in the study, both mice and humans, showed higher levels of ATF4. High ATF4 levels were also associated with increased recurrence after lung surgery and poorer survival among patients with lung adenocarcinoma, the most common form of lung cancer.
“Our results suggest that ATF4 is not only part of the mechanism behind the spread of lung cancer but may also serve as a marker of more aggressive disease,” says another author.
The study opens the door to a treatment strategy targeting the age-related signaling system that the tumor has hijacked. By blocking ATF4, or a specific metabolic process controlled by ATF4, with drugs, the researchers were able to dramatically reduce the spread of old tumors in mice.
Previously, it has been unclear why studies using similar drugs have largely failed when tested in humans. The new findings suggest that these treatments may need to be targeted more precisely to the right patient groups. ScienceMission sciencenewshighlights.